|
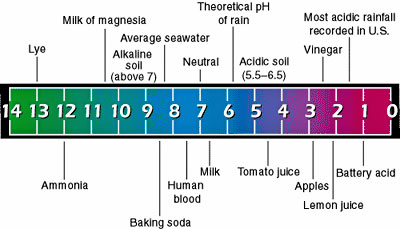
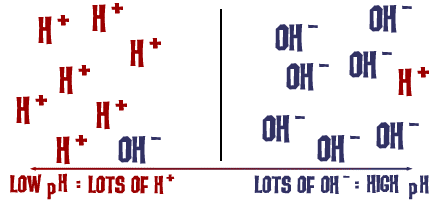
We receive many questions about PH and Alkalinity. It is important to know about both in order to maintain your spa effectively. If the PH and/or Alkalinity get out of whack then the water can become too basic or, even worse, too acidic. It is rare for water to become too basic (usually it involves some kind of cleaning agent that hasn't been washed away thoroughly) but we encounter acidic water much too often. To understand these concepts, we must first understand PH. PH, in its simplest form, is the measure of H+ ions in an aqueous solution. Acids, which lower the PH of a solution, donate an H+ ion (known as a proton). The lower the PH of a solution, the higher the number of these H+ ions are in a given amount of water. Common acids include Citric Acid (in lemons), Hydrochloric Acid (in our stomach and in pool cleaner), and Acetic Acid (in vinegar). These H+ ions can cause problems in a spa because they easily react with, dissolve, or degrade the materials used in spa equipment. Bases, on the other hand, produce HO- (Hydroxide) ions. While high concentrations of these can also damage spa parts, it requires a strongly caustic chemical to produce these concentrations. These HO- ions react with the H+ ions, neutralizing them and producing pure water. Since HO- ions neutralize H+ ions, there will be much less H+ in a basic solution. Since pure water is perfectly neutral, any solution with a concentration of H+ less than that in pure water is considered basic. The ideal PH of a spa is between 7.2 and 7.8. This prevents bacteria growth, is the best for your skin, and preserves spa equipment. Alkalinity is very similar to PH, but differs in some important ways. Alkalinity can be described as the ability of a solution to neutralize H+ ions (acidity) and resist changes to PH. Specifically, Alkalinity usually refers to the amount of dissolved carbonate (a weak base) in water. While this dissolved carbonate doesn't make the water very basic, it does stop the water from becoming acidic when an acid is introduced. The higher the Alkalinity in your spa, the more acids (such as the ones on your skin) can be added to the water without lowering the PH. So Alkalinity is basically (get it, BASICally?) the ability of the water to resist PH changes.
Now remember, too much Alkalinity means some of the dissolved carbonates can build up in the pumps/motors. The ideal range is about 80-120 parts per million in spa water. This is high enough to prevent drastic PH changes, while low enough to prevent carbonate buildup.
0 Comments
Leave a Reply. |
Details
AuthorLee Glunt has been in the Spa business pretty much since birth. Feel free to call or text with any questions. Archives
September 2016
|